- Author Arianna Cook cook@women-community.com.
- Public 2023-12-17 14:23.
- Last modified 2025-01-23 09:40.
The fight against illegal drugs in competitions has been going on for a very long time. Find out how steroids are calculated in the blood of Olympic athletes. Most people believe that doping began to be used in sports after the creation of the first AAS. However, archaeologists have found references to the fact that Philostratus and Galen also describe attempts by athletes to increase strength and endurance at the Olympic Games held in Ancient Greece. For this they used decoctions of seeds of various plants and fungi.
In ancient Rome, owners of racehorses turned to similar tricks, giving them a special drink that was supposed to increase their strength. In every era, people wanted to become stronger and faster, using various drugs for this. Today we will talk about doping control methods in Olympic sports.
Method # 1: Gas chromatography
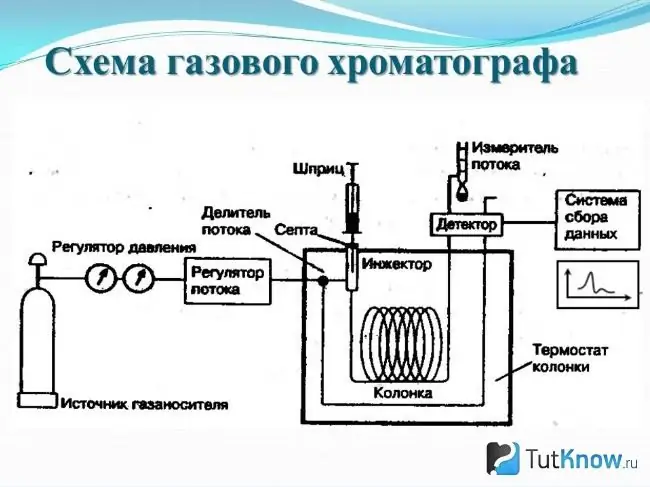
Capillary columns have become the most popular gas chromatography tool for doping testing today. They are actively used in carrying out a complete analysis or in the search for a specific substance. The column consists of the following main parts:
- External protective coating;
- Sorbent layer;
- Stationary phase.
Sorbent layer
This layer is made of high purity synthetic quartz glass. Since this material contains silanol groups, its surface is highly active and can interact with certain groups of the analyte, for example, hydroxyl, thiol residues, etc. As a result, peaks of substances to be separated appear on the surface of the sorbent layer. Before use, the sorbent layer is subjected to appropriate chemical cleaning and only after that the stationary phase is applied to it.
Stationary phase
In this method of doping control, the stationary phase is of great importance. Thanks to it, it becomes possible to determine the retention time, the quality of separation and the firm of the peaks of the analyte. The stationary phase is a special part of the capillary columns and is made from a specific type of material. Most often it is a substituted polysiloxane with a high resistance index.
The number and structure of substituted groups is the main characteristic of the stationary phase. However, there is also a significant drawback in the stationary phase, namely, high sensitivity to oxygen. This leads to phase destruction at high temperatures.
Outer shell
Capillary columns are fragile and therefore need protection. Most often, the outer shell is made of polyimide. This makes the columns strong enough and, when the outer shell is applied, the polyimide fills in all microdefects, stopping their further development.
Method # 2: Liquid chromatography
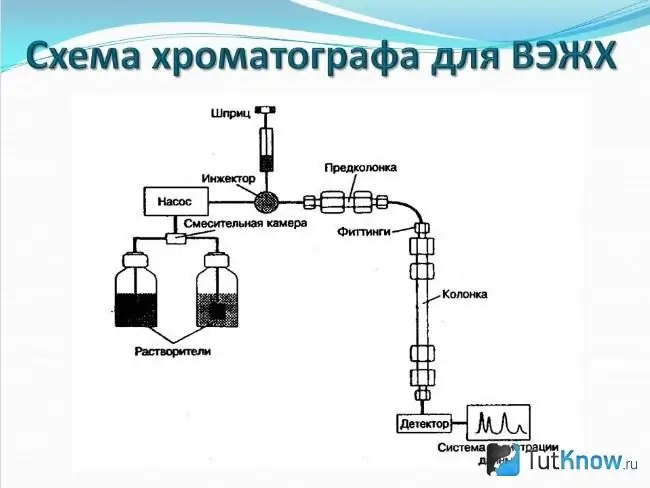
Compared to the previous doping control method, liquid chromatography has a fairly wide variety of fillers and sizes. It should also be said that when using this method, it is possible to use several methods for separating substances.
Instead of capillary columns, this method uses cartridges. Today, thanks to the improvement of technologies, it has been possible to significantly reduce their size and at the same time increase productivity.
When using any chromatographic method, the stationary phase is important. When selecting it, a large number of factors are taken into account, for example, the size of the investigated particles or the characteristics of the carrier.
Method # 3: Detectors

The detection and identification of substances separated by chromatography during doping control is of particular importance. A large number of all kinds of systems are in use now. It makes no sense to describe everything, but a few of them can be described in more detail.
Plasma ionization detector
This device is used in gas chromatography and can be called the most versatile among all existing ones. Leaving the capillary column, the gas mixes with air, which contains a large amount of hydrogen. The resulting mixture then ignites. After the combustion of hydrogen, a certain amount of ions of this substance remains in the air.
However, during pyrolysis, various organic substances also form electrons and ions, which significantly increases conductivity. When voltage is applied to the collecting electrode, an electric current appears, the strength of which is proportional to the amount of the sample under study, which burns out after leaving the capillary column. After that, it remains only to measure the current strength using an ammeter.
You will learn about doping control in Olympic sports from this story:
[media =






